PRODUCT IDENTIFICATION
H.S. CODE
TOXICITY
PRICE
U$25/kg CFR by sea for 100kgs
CLASSIFICATION
PHYSICAL AND CHEMICAL PROPERTIES
AUTOIGNITION
NFPA RATINGS
REFRACTIVE INDEX
GENERAL DESCRIPTION & APPLICATIONS
Monochloroacetic acid, chlorinated simplest carboxylic acid, has electron-withdrawing atoms on the next carbon to acid. Alha-chlorine makes monochloroacetic acid more acidic than acetic acid. The more electronegative atom presence tends to strengthen the acidic property.
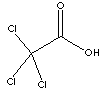
Trichloroacetic acid is stronger than monochloroacetic acid. Trichloroacetic acid is a corrosive and toxic crystals, with pungent odour; boiling point 198 C and melting point 58 C. It is soluble in water and the solution is a strong acid. Trichloroacetic acid can be used as a useful chemical intermediate for the production of various target molecules (medicine, pharmacy, and herbicides). The sodium salt is used as a weedkiller directly. It is used as reagent for albumin detection.
Dichloroacetic acid is a colourless to slightly yellowish liquid, highly corrosive. It is miscible with water and is readily soluble in alcohols, ketones, hydrocarbons and chlorinated hydrocarbons. It is typically prepared by the reduction of trichloroacetic acid. It has shows the weaker acidity than benzenesulphonic acid or trichloroacetic acid, which provides milder reaction conditions for detritylation in the oligonucleotide synthesis and more stability against decomposition to hydrochloric acid. It is used as an intermediate in the production of glyoxylic acid, dialkoxy and diaroxy acids, and sulfonamides. Some end products include chloramphenicol and thiamphenicol. It is used in preparations of iron chelates for agricultural industry. It is used to a lesser extent as a cauterizing agent in dermatology.APPEARANCE
ASSAY
Cl
0.001% max
HEAVY METALS
20ppm max
25kgs in plastic drum